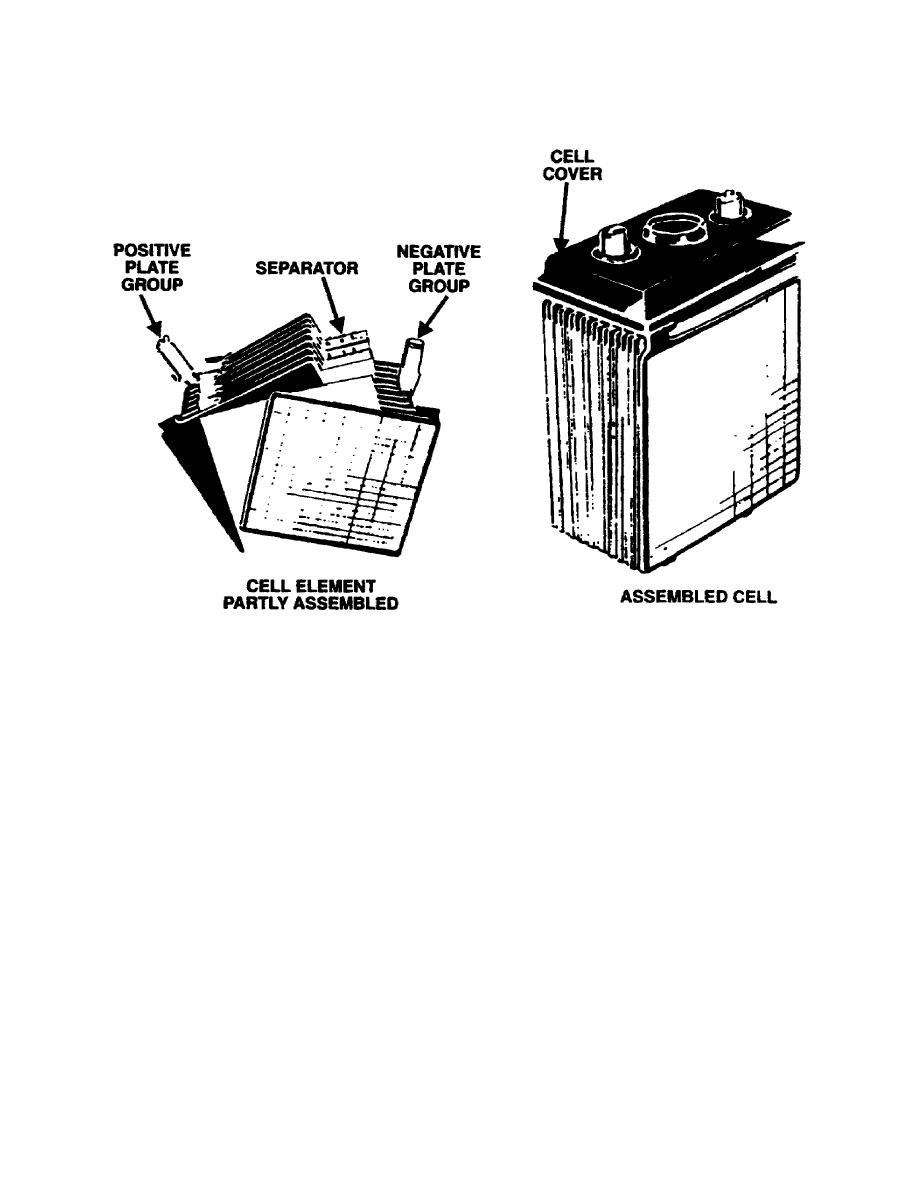
An example of a secondary cell is shown in the drawings below. The positive electrode is
made up of the positive plate group and the negative electrode of the negative plate group.
Figure 1-4.
The positive plate group makes up the positive electrode. The negative plate group makes up
the negative electrodes. Placing the assembled cell in a container of electrolyte will cause a
chemical action that will generate electricity. A secondary cell differs from a primary cell in
that a secondary cell can be recharged and a primary cell can only be restored.
1-5
IT0335




 Previous Page
Previous Page
