Decreases.
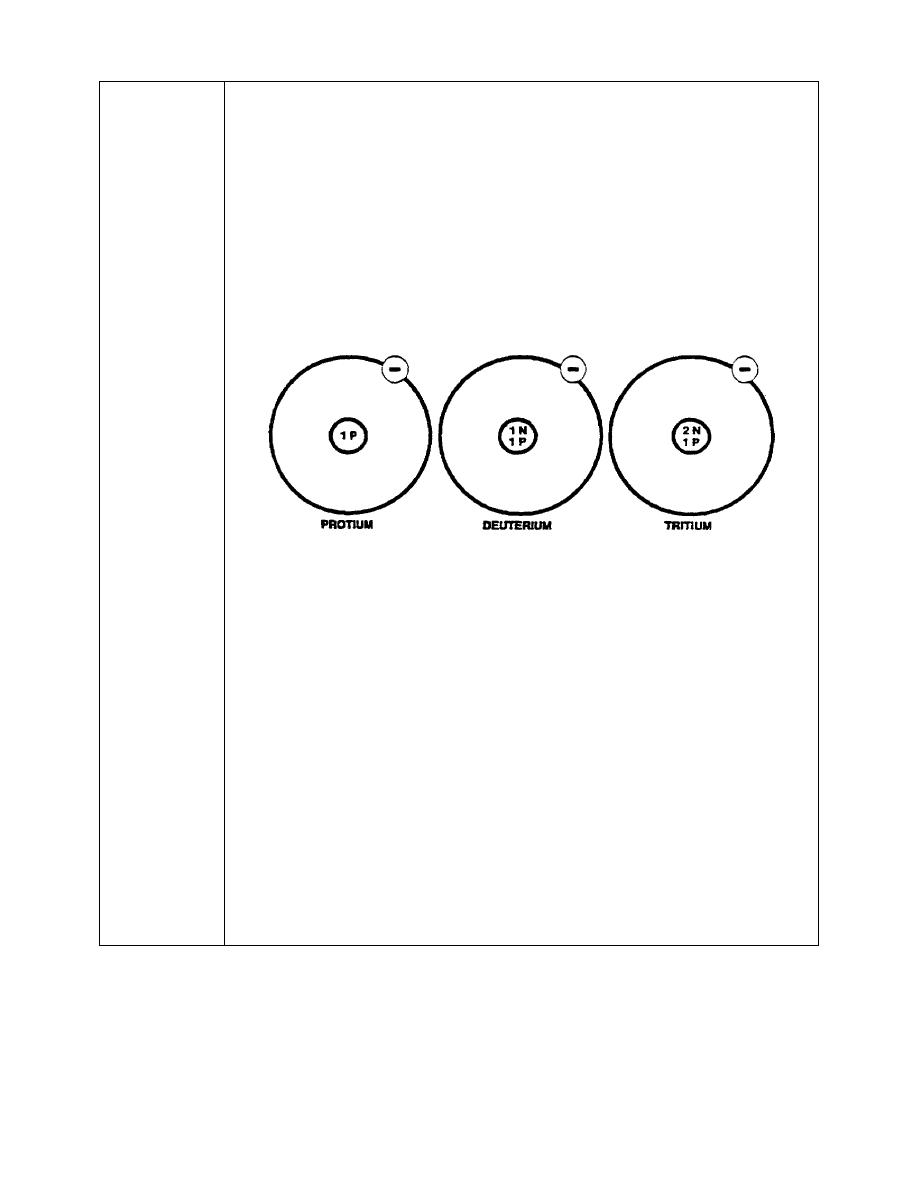
37. A large number of elements exist in two or more forms called isotopes.
decreases All the atoms of an element share the common feature of having
the same number of protons; thus, they all have the same atomic number.
The isotopes, or different forms of the same element, differ only by the
isotopes, which are shown below.
Since the protons and the neutrons make up the largest part of the atom,
each of the different isotopes of an element has a different atomic mass.
mass of one neutron.
Isotopes are different forms of an element, each having a different atomic
___________________ ,
1-27
IT0341


 Previous Page
Previous Page
